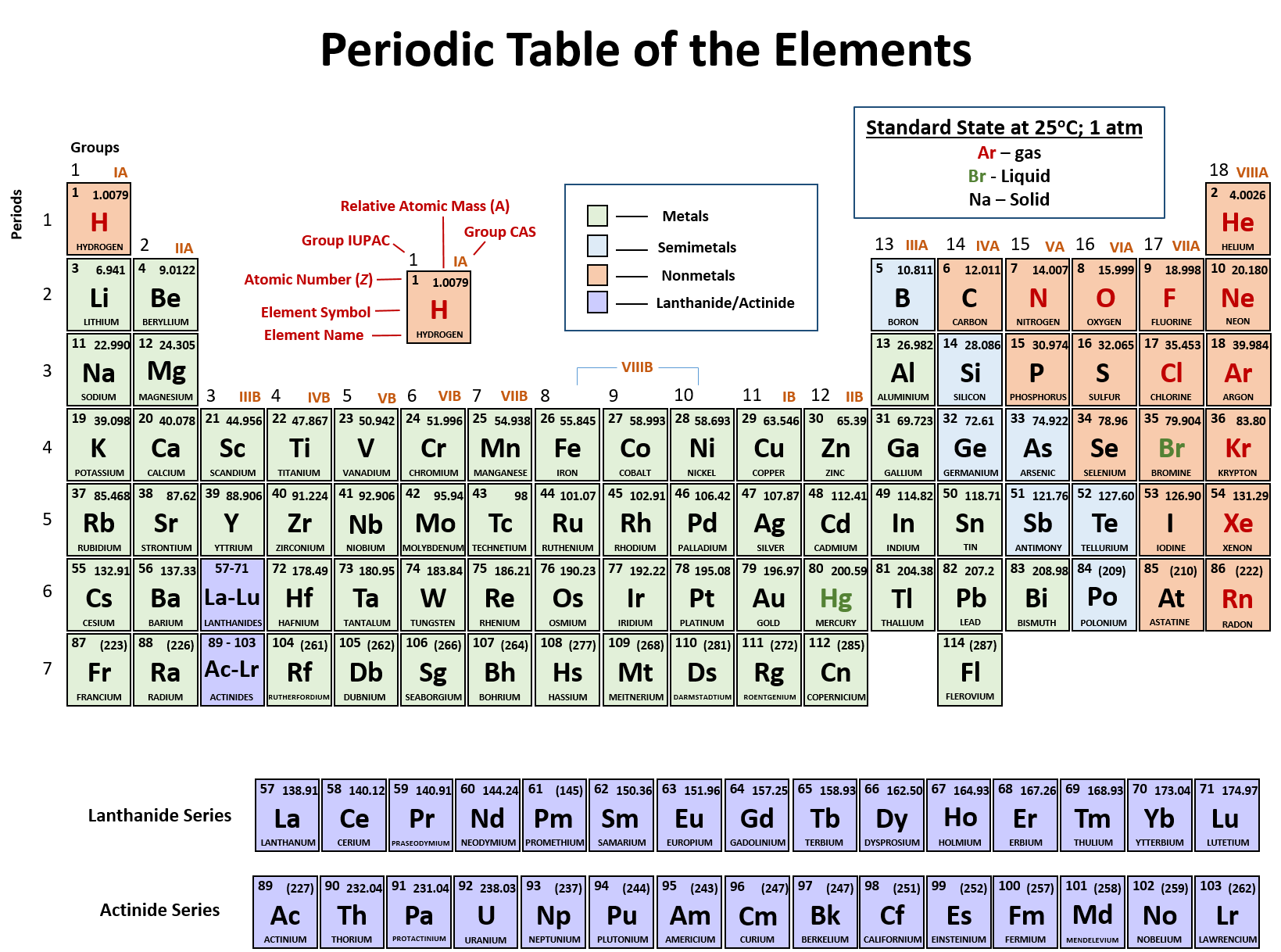
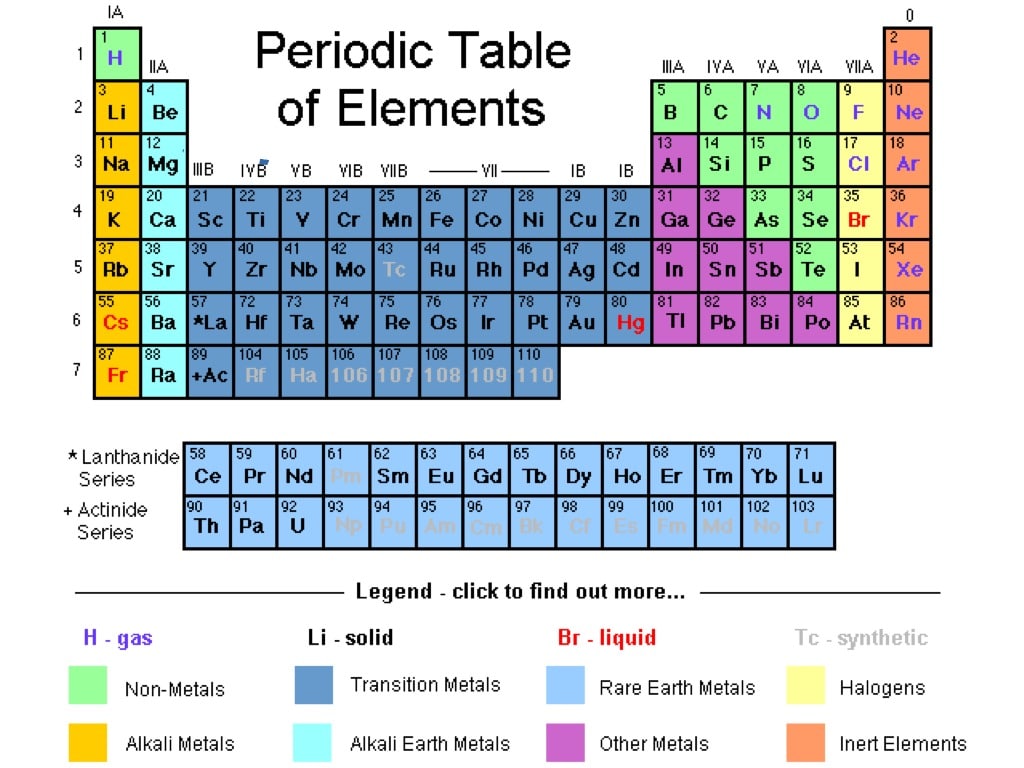
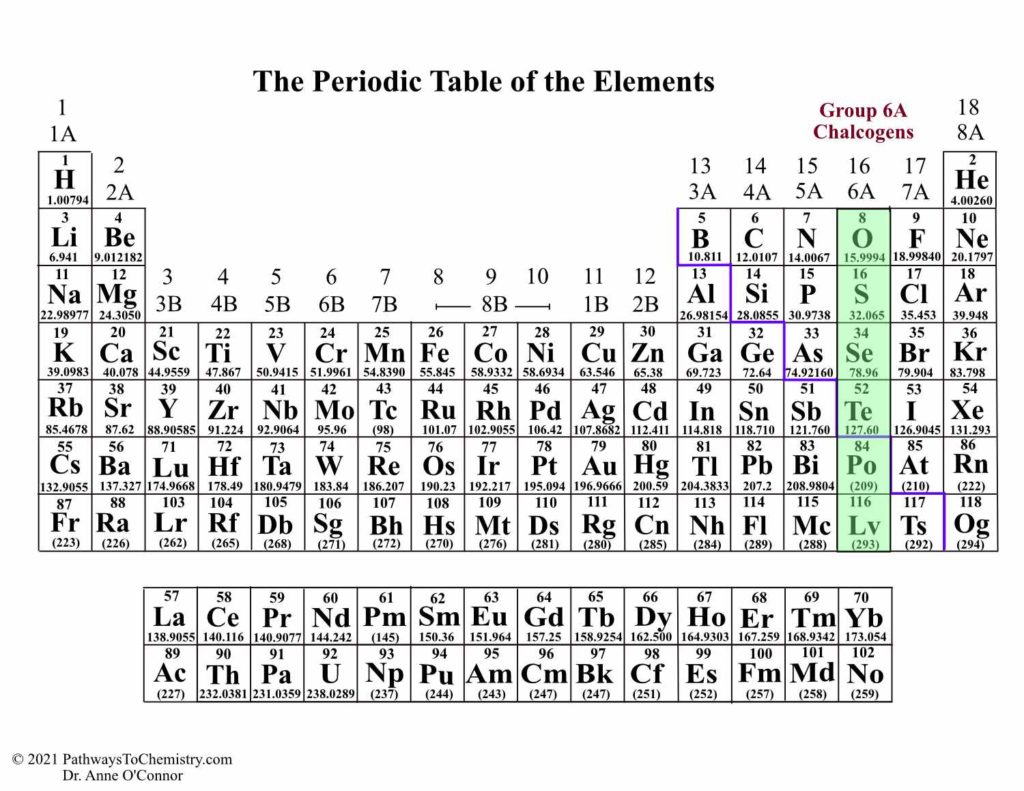
 He left gaps in his table for what he thought were undiscovered elements, and he made some bold predictions regarding the properties of those undiscovered elements. Later that decade, Dmitri Mendeleev, a Russian chemist, organized all the known elements according to similar properties. In 1864, Julius Lothar Meyer, a German chemist, organized the elements by atomic mass and grouped them according to their chemical properties. Likewise, lithium, sodium, and potassium react with other elements (such as oxygen) to make similar compounds. For example, chlorine, bromine, and iodine react with other elements (such as sodium) to make similar compounds. ^ IUPAC Provisional Recommendations for the Nomenclature of Inorganic Chemistry (2004) (online draft of an updated version of the " Red Book" IR 3-6.In the 19th century, many previously unknown elements were discovered, and scientists noted that certain sets of elements had similar chemical properties.Sometimes the group number is used, as in group 14 element, and sometimes the name of the first element in the group is used, as in carbon group or carbon family. Heavy atom - term used in computational chemistry to refer to any element other than hydrogen and helium.Īny periodic table group can also be used in this sense.Superactinide - Hypothetical series of elements 121 to 153, which includes a predicted "g block" of the periodic table.Minor actinide - Actinides found in nuclear fuel, other that U and Pu: Np, Am, Cm.Transplutonium element - Elements with atomic number greater than 94.Transactinide element - Elements after the actinides (atomic number greater than 103).Transuranium element - Elements with atomic number greater than 92. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
